Get Ahead with Grant Opportunities for Optics & Photonics
Ways to Stretch Marketing Budgets with Grant Funding In a time of economic uncertainty, you need marketing more than ever to hit your goals, but like...


For optics and other component manufacturers, medical devices represent a strong opportunity—especially for those who need to diversify their revenue base from DoD.
The US medical device market growth rate has slowed in recent years; it’s projected at 6.1% for 2017. There are several factors slowing growth and innovation in US healthcare, including:
If you’re an OEM or contract manufacturer selling into the medical device market and your business plan calls for double digit growth, you’ll have to compete for it.
While overall growth is slowing, McKinsey shows some good news for optics and photonics: acceptance and growth of medical devices that rely on imaging. Many of the companies we work with will be involved in products that are increasingly relying on innovative optical systems to reduce size and cost while increasing performance.
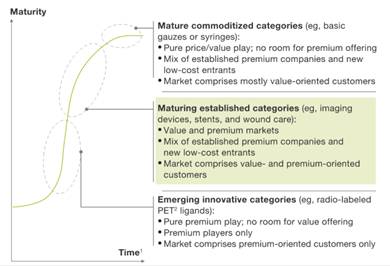
New to the market? Your value proposition for medical devices will differ significantly to the other markets you serve. This impacts your key messages, the images you use, and other aspects of your branding and marketing.
Every industry has its “magic words,” but you have to back them up with real data. An effective inbound marketing program will catch prospects’ attention, bring them a little deeper, back it up with data, and inspire them to take action. Think of it like an onion: allow the prospect to pull back as many layers as they’d like.
The right “hook” is dependent on your target customer persona, and it will be different company to company, but here are some proven marketing messages for medical device companies:
There is a tremendous amount of regulation and rigor involved in purchasing decisions in this market. It is not a decision made on the golf course. Expect ISO certification requirements and audits, and making significant investments in:
Some of the companies we’ve worked with have had success with technical content development, for example:
Download our White Paper Writing Guide to start creating your own technical content.
If you want to develop a sales and marketing strategy to increase your revenue base in the medical device market, then your first key customer success is critical. Consider a pilot test, STTR, or SBIR to establish credibility and publishable proof points.
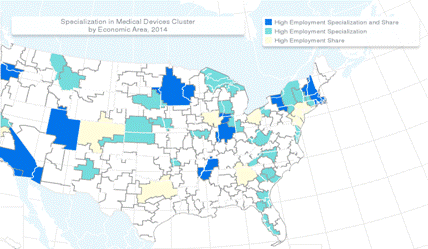
While the most effective sales territories are based on industry expertise, not geographies, clustering can be useful to maximize return on sales travel. When you’re visiting a client or attending a conference, seek out prospects in the same geography. Target clusters for highest sales productivity and service efficiency.
Looking to resonate with medical device customers? Check out our medical device series to learn customers’ top of mind concerns and challenges and how to create a value proposition that wins.

Ways to Stretch Marketing Budgets with Grant Funding In a time of economic uncertainty, you need marketing more than ever to hit your goals, but like...

Increasing brand awareness is a top goal for B2B companies in 2025. But what is a brand? And how can you evolve it to meet your changing company...

Why Even Innovation Powerhouses Struggle to Launch Breakthrough Products (and How to Fix It) In recent years, product launches have slowed. Even...